In summary, treatment of patients with suspected NP starts with immediate initiation of antibiotics and collection of respiratory secretions. While low risk patients can receive narrower spectrum therapy, most patients will require broad initial empiric therapy. The antibiotic regimen, however, should be narrowed at 48–72 hours based on microbiological results if the patient is improving. Overall treatment duration of 1 week is safe and effective with less chance of promoting growth of resistant organisms. In the subset of patients with pseudomonal infections, treatment of 1 week duration should be followed by active surveillance for recurrence, or alternatively, treatment can be extended to two weeks.
Prognosis
Once treatment for NP is initiated, clinical improvement is usually seen by 48–72 hours. There is little support for following either microbiologic response (clearance of positive cultures) or the response by chest radiography. The chest radiograph often lags behind the clinical response, however, a markedly worsening CXR (>50% increase in infiltrate) within the first 48 hours may indicate treatment failure. Clinical resolution as measured by temperature, white blood cell count, and oxygenation usually occurs by 6–7 days (10). Failure of oxygenation to improve by 72 hours has been shown to be predictive of treatment failure.
The overall mortality in patients with NP is as high as 30–70%, largely due to severe comorbid disease in the at risk population. Higher mortality rates are seen in patients with VAP and resistant organisms. The mortality attributable to the episode of NP is about 30%, and can be reduced to <15% with appropriate antibiotic therapy (1).
Prevention
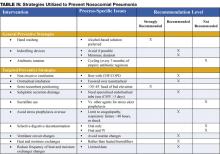
Preventive strategies are either directed at reducing the overall incidence of infectious complications in hospitalized patients, or they are specifically targeted at reducing the incidence of nosocomial pneumonia (3). The majority of the data supporting preventive strategies is limited to patients in the ICU, and in particular, patients receiving mechanical ventilation. However, many of the preventive principles can be extrapolated to the non-ICU population. The preventive strategies are highlighted in Table IV (page 18).
General Preventive Strategies
General preventive strategies aim to avoid contamination of patients with antimicrobial resistant organisms that exist in hospitals, or mitigating the emergence of antimicrobial resistant organisms in the first place. Preventing iatrogenic spread of resistant organisms depends on careful hand hygiene. Hand washing before and after patient contact reduces the incidence of nosocomial infection. Alcohol-based hand rinses placed at the bedside may actually be superior to soap and water, and in addition, improve compliance with hand hygiene.
Minimizing the use of indwelling devices (central lines, urinary catheters) also reduces the emergence of resistant organisms. When these devices are necessary, focusing on their timely removal is critical. The control of antibiotic use has been central to many preventive strategies. Prolonged or unnecessary use of broad-spectrum antibiotics is strongly associated with development and colonization of resistant organisms. Strategies that focus on aggressive antibiotic de-escalation (described above) are a key preventive tool. Some institutions have had success with antibiotic restriction or rotation, but long term data on the effectiveness of these techniques are lacking.
Targeted Preventive Strategies
Preventive strategies to lower the incidence of NP focus on reducing risk factors for oropharyngeal or gastric colonization and subsequent aspiration of contaminated oropharyngeal or gastric secretions (1,3,7,11).
Endotracheal intubation is one of the most important risk factors for NP in patients requiring ventilatory support. The use of non-invasive ventilation (NIV) or positive pressure mask ventilation in selected groups of patients has been effective in preventing nosocomial pneumonia. Non-invasive ventilation has been most successful in patients with acute exacerbations of chronic obstructive pulmonary disease (COPD) and pulmonary edema secondary to congestive heart failure (CHF) and should be considered in appropriately selected patients. When intubation is required the use of nasotracheal intubation should be avoided due higher rates of NP when compared to orotracheal intubation.