Case
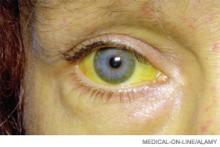
A 56-year-old man with a history of cirrhosis, complicated by esophageal varices and ongoing alcohol abuse, is admitted after his wife found him lethargic and disoriented in bed. His wife said he’d been increasingly irritable and agitated, with slurred speech, the past two days. On exam, he is somnolent but arousable; spider telangiectasias and asterixis are noted. Laboratory studies are consistent with chronic liver disease.
What is the best therapy for his acute hepatic encephalopathy?
Overview
Hepatic encephalopathy (HE) describes the spectrum of potentially reversible neuropsychiatric abnormalities seen in patients with liver dysfunction. The wide range of neuropsychiatric presentations led to the development of consensus HE classification terminology by the World Congress of Gastroenterology in 2002.
The primary tenet of all HE pathogenesis theories is firmly established: Nitrogenous substances derived from the gut adversely affect brain function. These compounds access the systemic circulation via decreased hepatic function or portal-systemic shunts. In the brain, they alter neurotransmission, which affects consciousness and behavior.
HE patients usually have advanced cirrhosis and, hence, many of the physical findings associated with severe hepatic dysfunction: muscle-wasting, jaundice, ascites, palmar erythema, edema, spider telangiectasias, and fetor hepaticus. Encephalopathy progresses from reversal of the sleep-wake cycle and mild mental status changes to irritability, confusion, and slurred speech.
Advanced neurologic features include asterixis or tongue fasciculations, bradykinesia, hyperreflexia, and ultimately coma. History and laboratory data can reveal a precipitating cause (see Table 2, p. 19). Measurement of ammonia concentration remains controversial. The value may be useful for monitoring the efficacy of ammonia-lowering therapy, but elevated levels are not required to make the diagnosis.
Multiple treatments have been used to manage HE, yet few well-designed randomized trials have assessed efficacy due to challenges inherent in measuring the wide range of neuropsychiatric presentations. Nonetheless, a critical appraisal of available data delineates a rational approach to therapy.
Review of the Data
In addition to supportive care and the reversal of any precipitating factors, the treatment of acute HE is aimed at reducing or inhibiting intestinal ammonia production or increasing its removal (see Table 1, left).
Nonabsorbable disaccharides (NAD): Lactulose (beta-galactosidofructose) and lactitol (beta-galactosidosorbitol) are used as first-line agents for the treatment of HE and lead to symptomatic improvement in 67% to 87% of patients.1 They reduce the concentration of ammoniogenic substrates in the colonic lumen in two ways—first, by facilitating bacterial fermentation and secondary organic acid production (lowering colonic pH) and, second, by direct osmotic catharsis.
NAD are administered orally or via nasogastric tube at an initial dose of 45 ml, followed by repeated hourly doses until the patient has a bowel movement. For patients at risk of aspiration, NAD can be administered via enema (300 ml in 700 ml of water) every two hours as needed until mental function improves. Once the risk of aspiration is minimized, NAD can be administered orally and titrated to achieve two to three soft bowel movements daily (the usual oral dosage is 15 ml to 45 ml every eight to 12 hours).