Sickle cell anemia is a prototypical single gene deletion disorder familiar to medical students everywhere. Most physicians recall that it is a devastating disorder that starts with a single substitution of valine for glutamic acid in the beta-globin gene of the hemoglobin molecule, rendering the hemoglobin molecule unstable in its de-oxygenated state. This leads to polymerization of hemoglobin molecules within the red cell, deformation of the cell membrane, and sickling. Sickling, in turn, causes blood vessel damage, vaso-occlusion, and various other physiologic effects, which lead to both micro- and macro-vascular protean complications.1
Despite extensive basic research in sickle cell disease (SCD), clinical research into the optimal management of this complex disease has lagged behind. Many adult hospitalists may be unfamiliar with the care of adult SCD patients because most of these patients are cared for in academic centers by hematologists.
In this article we review the common complications of sickle cell anemia in adult patients, the management of associated conditions, and the evidence base for treatment guidelines.
Overview
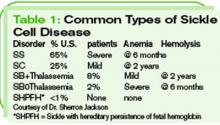
Sickle cell anemia is not just one disorder, but is, rather, a collection of related disorders involving mutations of the hemoglobin molecule. (These disorders are categorized in Table 1, p. 40) In the United States, SCD primarily affects black Americans, with about 9% of this population having sickle trait.2 One in 600 black Americans has sickle cell anemia, also known as hemoglobin SS disease, and there are an estimated 72,000 patients with SCD in the United States.2
In the early 1970s, the average life expectancy for patients with SCD was estimated to be 14.3 years.3 At that time, more than half of SCD patients died before age five, primarily from infectious complications such as pneumococcal sepsis. Twenty years later, the Cooperative Study of SCD documented a much better life expectancy: an average of 42 for males and 48 for females with homozygous S disease (the set of patients with two copies of the defective hemoglobin S mutation).
On average, patients with hemoglobin SCD routinely survive into their 60s. Many factors, including earlier diagnosis of SCD through universal screening programs, better vaccines and vaccination rates, prophylactic antibiotics in infancy and young childhood, and aggressive treatment of fever in infants, as well as other advances in care, have contributed to this progress. A longer life expectancy in this population has heralded the extension of common complications into adulthood, however, along with the emergence of other adult-specific complications of SCD. (See Table 2, p. 40.) In order to best treat adults with SCD, hospitalists should be aware of these complications, as well as of the advances that have been achieved.
Extension of Pediatric Complications
Perhaps the most common manifestation of SCD in children and adults is the painful crisis. This manifests in infants as a painful swelling in the digits of the hands and feet known as dactylitis. In older children and adults, pain occurs more often in the long bones of the arms and legs and in the sternum, vertebrae, and pelvic bones. Risk factors for painful crises include higher hematocrit values and higher sickle fractions, typically greater than 30%.4 Frequent painful crises are a marker of disease severity and an independent risk factor for death in SCD.
Hospitalists who regularly admit SCD patients are familiar with a subset of “frequent flyers” who experience recurrent painful crises. Remember that this is a small fraction of the total SCD population. In fact, 40% of SCD patients don’t suffer any painful crises requiring medical attention in a given year, and only 1% face more than six such events.5 Pain management in SCD is perhaps beyond the scope of this article, and there are no widely accepted guidelines. A few expert reviews are available for guidance, however.6,7