Despite repeated warnings for more than 25 years by the Institute for Safe Medication Practices (ISMP) and other organizations, one of the major causes of medication errors is the ongoing use of potentially dangerous abbreviations and dose expressions. Root cause analyses information contained in the Joint Commission Sentinel Event Database shows that the underlying factors contributing to many of these medication errors are illegible or confusing handwriting by clinicians and the failure of healthcare providers to communicate clearly with one another.
Symbols and abbreviations are frequently used to save time and effort when writing prescriptions and documenting in patient charts; however, some symbols and abbreviations have the potential for misinterpretation or confusion. Examples of especially problematic abbreviations include “U” for “units” and “µg” for “micrograms.” When “U” is handwritten, it can often look like a zero. There are numerous case reports where the root cause of sentinel events related to insulin dosage has been the interpretation of a “U” as a zero. Using the abbreviation “µg” instead of “mcg” has also been the source of errors because when handwritten, the symbol “µ” can look like an “m.” The use of trailing zeros (e.g., 2.0 versus 2) or use of a leading decimal point without a leading zero (e.g. .2 instead of 0.2) are other dangerous order-writing practices. The decimal point is sometimes not seen when orders are handwritten using trailing zeros or no leading zeros. Misinterpretation of such orders could lead to a 10-fold dosing error.
A New Approach
As part of efforts to improve patient safety, the Joint Commission has long worked with hospitals to develop practical, cost-effective strategies that can be implemented at organizations regardless of unique characteristics, such as ownership, size, or location. One such Joint Commission initiative is a National Patient Safety Goal to improve communication. This goal and one of its requirements specifically addresses the role that abbreviations, acronyms, symbols, and dose designations play in medication errors.
The Joint Commission began establishing National Patient Safety Goals in 2002 as a means to target critical areas where patient safety can be improved through specific action in healthcare organizations. The resulting National Patient Safety Goals are designed to give focus to evidence-based or expert consensus-based, well-defined, practical, and cost-effective actions that have potential for significant improvement in the safety of individuals receiving care. New Goals are recommended annually by the Sentinel Event Advisory Group, a Joint Commission-appointed, multidisciplinary group of patient safety experts.
JCAHO Expectations
In order to comply with the National Patient Safety Goal related to abbreviations, an organization must conduct a thorough review of its approved abbreviation list and develop a list of unacceptable abbreviations and symbols with the involvement of physicians. In addition, organizations must do the following to meet this goal:
- The list of prohibited abbreviations, acronyms, symbols, and dose designations must be implemented for all handwritten, patient-specific communications, not just medication orders;
- These requirements apply to printed or electronic communications;
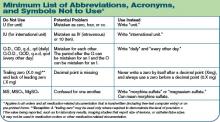
- This goal requires organizations to achieve 100% compliance with a reasonably comprehensive list of prohibited dangerous abbreviations, acronyms, symbols, and dose designations. This list need not be as extensive as some published lists, but must, at a minimum, include a set of Joint Commission-specified dangerous abbreviations, acro-nyms, symbols, and dose designations (see “Minimum List of Abbreviations, Acronyms, and Symbols Not to Use,” top right) and
- An abbreviation on the “do not use” list should not be used in any of its forms—uppercase or lowercase, with or without periods.