Case Presentation
A 68-year-old man presented to a university hospital with a 4-day history of sudden, progressive finger ischemia. His past medical history was significant for type II diabetes mellitus and hyperlipidemia. He also suffered from severe vascular disease. Four years prior to admission, he underwent several surgeries, including right carotid endarterectomy, coronary artery bypass, and right-lower-extremity revascularization. One year prior, he also required a left below-the-knee amputation due to vascular insufficiency. Additional history revealed long-standing asthma, hearing loss due to chronic bilateral otitis media, and multiple sinus surgeries in attempts to relieve recurrent infections. He also had lower-extremity peripheral neuropathy, attributed to diabetes and frequent steroid use for asthma control.
On admission, vital signs were stable. Physical exam demonstrated mild cyanosis of digits 2 through 5 on both hands. There were also scattered splinter hemorrhages and petechiae on the involved fingers. Rales were noted in the left lung base with diffuse wheezes. Cardiac and vascular exams were unremarkable. Chronic ulceration of the right toes was also noted. Laboratory studies were significant for a white blood cell count of 26,700 cells/mL with 52% eosinophils and a positive perinuclear antineutrophil cytoplasmic antibody (p-ANCA).
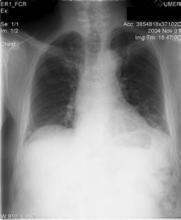
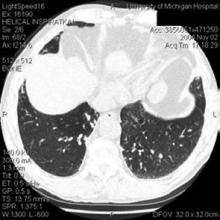
A chest radiograph revealed hazy, bilateral perihilar and left lower lobe infiltrates (Figure 1). Computed tomography of the chest was then performed and showed bronchiectasis of the left lung (Figure 2). This prompted a transbronchial biopsy that yielded tissue consistent with chronic inflammation.
What Is the Diagnosis?
Churg-Strauss Syndrome
Discussion
Churg-Strauss syndrome (CSS) is an allergic and granulomatous vasculitic illness affecting multiple organ systems. It typically follows 3 phases of progression. The first occurs in the second or third decade of life and includes the development of asthma in addition to chronic ear, nose, and sinus inflammation or infection. This is followed by eosinophilic infiltration of the lungs, skin, and other organs. The third phase commonly occurs 10 to 20 years after initial presentation, and it is heralded by small- and medium-vessel vasculitis.
In 1990, the American College of Rheumatology developed 6 diagnostic criteria for CSS and showed that having at least 4 of the 6 predicted the presence of CSS with a sensitivity of 85% and a specificity of 99.7%. They include:
- Asthma;
- Eosinophilia of >10% on a peripheral white blood cell count;
- Paranasal sinus disease;
- Mononeuropathy or polyneuropathy;
- Migratory or transient pulmonary opacities seen radiographically; and
- A blood vessel showing the accumulation of eosinophils in extravascular areas, as revealed by a biopsy.
Other helpful, but nonspecific diagnostic tests include a significantly elevated sedimentation rate, a positive p-ANCA with low titers of rheumatoid factor, high circulating IgE levels, and normocytic, normochromic anemia. CSS typically responds quite well to immunosuppressive therapy. The usual regimen consists of corticosteroids, and cyclophosphamide is frequently added. Before the advent of such therapies, CSS was consistently fatal, often within 3 months of the onset of vasculitis. Currently, 5-year survival rates exceed 70%.
In this patient, a diagnosis of CSS was based on history, clinical presentation, and laboratory results. Highdose methylprednisolone was initiated, and complete resolution of finger cyanosis and pain occurred in 48 hours. Oral cyclophosphamide was added the following day, and the patient was discharged home to complete 6 months of aggressive immunosuppressive therapy.
This presentation of CSS was rather unusual. Digital ischemia is uncommon in CSS, although it is consistent with the small-vessel vasculitis seen in this syndrome. Similarly, the late onset of the patient’s vasculitis is also unusual. The intermittent use of prednisone for asthma perhaps delayed the declaration of systemic symptoms.