Introduction
Clostridium difficile–associated diarrhea (CDAD) has been recognized with increased frequency as a cause of nosocomial illness. The frequency and incidence of CDAD varies widely, and is influenced by multiple factors including nosocomial outbreaks, patterns of antimicrobial use, and individual susceptibility. There are no reports of prospective studies by hospitals tracking positive toxin A or A/B and the outcomes of CDAD and its complications.
The Centers for Disease Control and Prevention (CDC) has analyzed secular trends in the incidence of CDAD, and it reported a steady increase from 1987 to 2001 (1). In this report, 30% of 440 infectious disease physicians who participated in a Web-based poll reported that they are seeing higher rates of CDAD, more severe CDAD, and more relapsing CDAD than in the past. There is an overall impression that there has been an increase in the proportion of cases with severe and fatal complications, and an increase in the relapse rate among affected patients.
In addition to morbidity and mortality, the economic burden of C. difficile infection in terms of delayed discharge and other hospital costs is considerable.
Epidemiology
The frequency and incidence of CDAD varies between hospitals and within a given institution over time. The risk for disease increases in patients with antibiotic exposure, gastrointestinal surgery, increasing length of stay in healthcare settings, serious underlying illness, immuno-compromising conditions, and advanced age.
C. difficile is shed in feces. Any surface, device, or material (e.g., commode, bathing tub, and electronic rectal thermometer) that becomes contaminated with feces may serve as a reservoir for C. difficile spores. Spores are transferred to patients mainly via the hands of healthcare personnel who have touched a contaminated surface or item (2-6).
The Organism and Pathophysiology of C. difficile Diarrhea
C. difficile is a gram-positive, anaerobic, spore-forming bacillus that is responsible for the development of antibiotic-associated diarrhea and colitis. C. difficile was first described in 1935 as a component of the fecal flora of healthy newborns and was initially not thought to be pathogenic (7). The bacillus was named difficile because it grows slowly and is difficult to culture. C. difficile is presently responsible for nearly all causes of pseudomembranous colitis and as many as 20% of cases of antibiotic-associated diarrhea without colitis. Although found in the stool of only 5% of the general population, as many as 21% of adults become colonized with this organism while hospitalized (2,6).
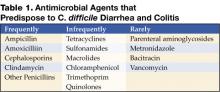
An alteration of the normal colonic microflora, usually caused by antibiotic therapy, is the main factor that predisposes to infection with C. difficile. Almost all antibiotics have been associated with C. difficile diarrhea and colitis. The antibiotics most frequently associated include clindamycin, cephalosporins, ampicillin, and amoxicillin (Table 1) (8).
In addition to antibiotic therapy, older age and severity of underlying disease are important risk factors for C. difficile infection. Other risk factors include the presence of a nasogastric tube, gastrointestinal procedures, acid antisecretory medications, intensive care unit stay, and duration of hospitalization (9).
C. difficile diarrhea is caused primarily by the elaboration of toxins A and B produced by bacterial multiplication within the intestinal lumen. These toxins bind to the colonic mucosa and exert their deleterious effects upon it. The organism rarely damages the colon by direct invasion, and diarrhea is caused by the effects of toxins produced within the intestinal lumen that adhere to the mucosal surface. Most toxigenic isolates produce both toxins, and about 5–25% of isolates produce neither toxin A nor B, and do not cause colitis or diarrhea (3-5).