The Case
A 62-year-old woman with a past medical history significant for metastatic adenocarcinoma of the lung presents to the ED with complaints of fever and shortness of breath. She has recently completed her first cycle of carboplatin, pemetrexed, and bevacizumab. Upon admission, she is found to have an absolute neutrophil count of 800 and a platelet count of 48,000. She is admitted for neutropenic fever and placed on IV antimicrobials. Sequential compression devices are initiated for DVT prophylaxis.
Key Clinical Questions
What risk do cancer patients have for VTE?
Patients with cancer have a risk of clinically significant VTE that is four to seven times that of patients without malignancy.1 This is due to a number of reasons:
- Tumor cells produce procoagulant activity inducing thrombin formation;2
- The cancer itself can compress or invade deep veins; and3
- Some cancer therapies such L-asparaginase and thalidomide/lenalidomide, plus high-dose steroids, or anti-estrogen medications such as tamoxifen can also increase patients’ risk of VTE.3,4,5
What inpatients with cancer need VTE prophylaxis?
Much like other hospitalized medical patients, patients with cancer who have reduced mobility and are not on therapeutic anticoagulation should receive pharmacologic prophylaxis unless there is a contraindication.3,6,7,8 Cancer patients with acute medical illnesses should also likely receive prophylaxis if there are no contraindications, because the vast majority of these have factors increasing their VTE risk, including infection, kidney disease, or pulmonary disease.3,6,7,8 Patients undergoing major cancer surgery should also receive pharmacologic prophylaxis prior to surgery and for at least seven to 10 days post-operatively.3,6,7,8
For ambulatory cancer patients who are admitted for short courses of chemotherapy or for minor procedures, however, there is not enough evidence to recommend routine VTE prophylaxis.6,7 An exception to this is patients with multiple myeloma receiving thalidomide-based or lenalidomide-based chemotherapy, who should receive pharmacologic prophylaxis.6,7
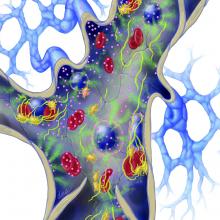
Image Credit: DNA ILLUSTRATIONS / SCIENCE SOURCE Illustration shows vascularized cancer cells in the background upper left, with arteries going into it and a venous plexus coming off which joins up with a bigger vein with valves. larger pink molecular balls represent the procoagulation factor that tumor cells produce that directly jump starts the coagulation pathway. the procoagulation factor molecules are binding to regular leukocytes, endothelium and platelets that will then start producing tissue factor that also encourages coagulation. Also shown are tumor cells binding to endothelium which cause production of tissue factor, causing clots to form. two of the clots are shown embolizing. fibrinogen and platelets are throughout.
What are the options available for VTE in hospitalized cancer patients?
The guidelines for VTE prophylaxis in hospitalized cancer patients recommend either unfractionated heparin (UFH) or low molecular weight heparin (LMWH) for prophylaxis when no contraindications exist.5 The only two LMWH that have been FDA approved for prophylaxis are enoxaparin and dalteparin. When deciding between UFH and LMWH, no evidence shows that one is better than the other in preventing VTE in hospitalized cancer patients.9 There is evidence that the use of LMWH results in a lower incidence of major hemorrhage when compared to UFH.10
What are the contraindications to pharmacologic VTE prophylaxis in cancer patients?
Contraindications for pharmacologic VTE prophylaxis in cancer patients include active major bleeding, thrombocytopenia (platelet count <50,000/µL), severe coagulopathy, inherited bleeding disorder, and at the time of surgery or invasive procedures (including lumbar puncture and epidural or spinal anesthesia).3,6,7 Those with contraindications to pharmacologic VTE prophylaxis should have mechanical prophylaxis instead.
What is the recommended treatment of VTE in cancer patients?
After the diagnosis of pulmonary embolism (PE) or DVT is found, LMWH is the preferred initial anticoagulant instead of UFH unless the patient has severe renal impairment (CrCl of less than 30 ml/min).6,7,8 LMWH is also preferred over warfarin for long-term anticoagulation during the initial six months of therapy.6,7,8 Following the initial six months, continued anticoagulation with either LMWH or warfarin could be considered in patients with active cancer, metastatic disease, or ongoing chemotherapy.6,7,8