Introduction
Urinary tract infections (UTIs) are serious bacterial infections and a common cause for hospital admission of infants and young children. The prevalence of UTI in infants younger than 1 year of age ranges from 3.3% to 6.5%, and between 1 and 2 years of age from 1.9% to 8.1%. Females outpace males across all age groups, with the exception of the first 3 months of life (1). Without appropriate treatment and management, UTI can result in dehydration, urosepsis, and long-term medical problems including hypertension, renal scarring, and decreased renal function. This review will focus on the inpatient management of first-episode UTI in infants and young children.
Diagnosis
Presenting symptoms in older children include urgency, frequency, dysuria, and complaints of back pain. In contrast, symptoms in infants and young children are often nonspecific and include irritability, diarrhea, vomiting, poor feeding, poor weight gain, crying on urination, and foul-smelling urine. The presence of a fever in infants and young children with UTI has been accepted as a marker of pyelonephritis, which occurs when infection has ascended to the upper collecting system of the kidney. Urinalysis (UA) and culture should be collected by suprapubic aspiration or transurethral catheterization, or by appropriately performed clean catch method for children of appropriate age and developmental ability. The use of a bag-collected urine specimen is insufficient and unreliable and should not be used in making the diagnosis of UTI. While suprapubic aspiration is considered the gold standard with a specificity and sensitivity of 100%, there is often resistance from parents and from physicians who are not properly trained to do this procedure.
The most accepted method of obtaining urine is sterile transurethral catheterization, results of which have 95% sensitivity and 99% specificity (2). When interpreting the UA, the most useful components for the diagnosis of a UTI include a positive leukocyte esterase, nitrite test, or gram stain on unspun urine, and microscopy revealing >10 white blood cells per high-powered field of spun urine. However, neonates under 30 days old may have no abnormality noted on initial UA (3,4). The presence of any bacteria on gram-stained urine offers the best sensitivity and specificity (5). Final diagnosis depends upon isolation of >105 of a single organism from a clean-catch specimen, or >104 of a single organism from a catheterized specimen.
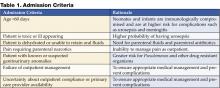
Admission Criteria
Guidelines for evaluation of serious bacterial infection and parenteral antibiotic use for febrile infants under 60 days of age should be followed. All febrile neonates less than 30 days of age should be admitted for parenteral antibiotics (6–11). Controversy exists on the need to use corrected or postconceptual age when evaluating and determining need for admission for febrile preterm infants, particularly for those under 35 weeks of gestation. Factors that can be considered by the practitioner include severity of Neonatal ICU course, severity of prematurity, and combined disease burden of UTI with common preterm comorbidities (anemia, apnea of prematurity, chronic lung disease).
Consider admitting and initiating parenteral antibiotic treatment using Table 1 as a guideline. Exercise a lower threshold for admitting infants and toxic-appearing young children due to concern for urosepsis, complications, and the need for appropriate and aggressive initial therapy.
Initial Inpatient Management
The 3 goals of inhospital treatment of UTIs are to effectively treat and eliminate the acute infection, prevent urosepsis in infants and immunocompromised children, and prevent and reduce long-term complications such as renal scarring, hypertension, and decreased renal function. Initial antibiotic treatment should be administered parenterally to ensure optimal antimicrobial levels and aimed at the most common organisms, including Escherichia coli, Klebsiella, Proteus, and Enterobacter spp. Less common organisms to consider include Pseudomonas, Enterococcus, Staphylococcus aureus, and group B Streptococcus. Organisms will differ on many factors, such as age, underlying disease, prior colonization, and antibiotic exposure.